Ceftriaxone + Sulbactam Combination for Effective Infection Control
Introduction to CEFORF-S Injection Range
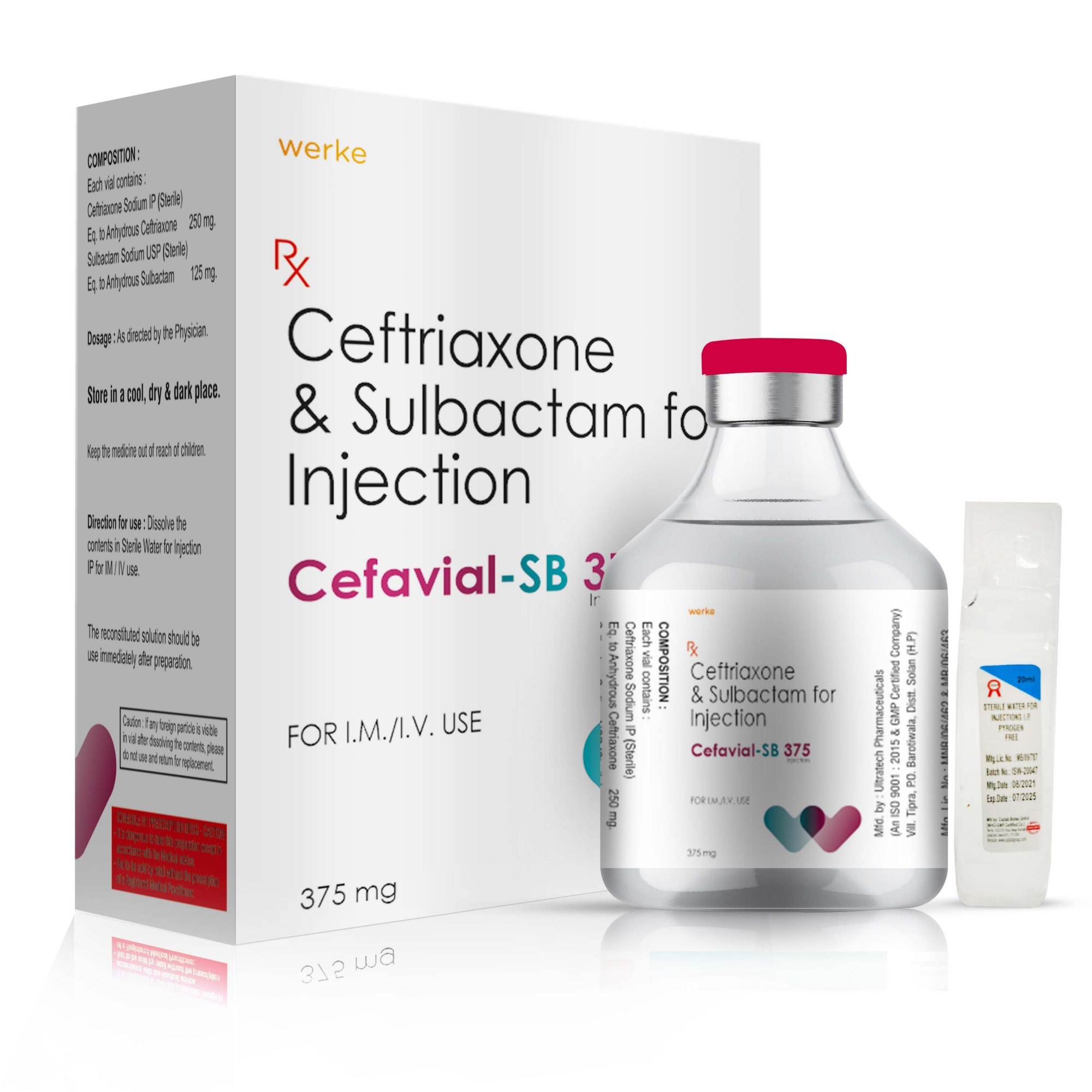
CEFORF-S offers potent antibacterial action, targeting a wide range of moderate to severe infections. Available in three strengths—375 mg, 750 mg, and 1.5 g—this formulation combines Ceftriaxone, a third-generation cephalosporin antibiotic, with Sulbactam, a beta-lactamase inhibitor. This synergy enhances the spectrum and potency of action, especially against resistant pathogens. CEFORF-S is trusted in clinical settings for treating respiratory, urinary, abdominal, and skin infections, as well as septicemia and bone infections. Its versatile IM/IV administration and multiple dosage options make it ideal for both adult and pediatric care, depending on severity and clinical judgment.
Composition and Strength Details
CEFORF-S injections are offered in three carefully calibrated strengths to cater to different levels of infection severity:
- CEFORF-S 375 mg
- Ceftriaxone 250 mg
- Sulbactam 125 mg
This lower dose is typically suited for mild to moderate infections and can be used in pediatric cases under medical supervision.
CEFORF-S 750 mg
Ceftriaxone 500 mg
Sulbactam 250 mg
Used for treating more advanced infections like bronchitis, pneumonia, or complicated UTIs. It offers an optimal combination of effectiveness and safety.
- CEFORF-S 1.5 g
- Ceftriaxone 1 g
- Sulbactam 500 mg
Recommended for serious infections such as meningitis, severe sepsis, or surgical prophylaxis in high-risk patients.
The combination works synergistically, where Sulbactam protects Ceftriaxone from degradation by beta-lactamase-producing bacteria, thereby restoring and extending its antibacterial activity. Its dual-action approach delivers wide-spectrum and sustained antibacterial coverage.
Recommended Dosage and Route of Administration
CEFORF-S can be administered either via intramuscular (IM) or intravenous (IV) injection, depending on the condition being treated and the urgency of action required.
The recommended adult dosage ranges from 750 mg to 1.5 g per day, given once or in split doses.
- Pediatrics: Dosage based on body weight; usually 50–75 mg/kg/day, under supervision.
Route:
- IM is often used for outpatient or mild cases.
- IV is preferred for rapid onset in hospitalized or severe infections.
Always reconstitute the powder using sterile water or compatible diluent as directed and administer under aseptic conditions. Renal or hepatic compromise should be considered when determining dosage.
Rare but Serious Effects to Monitor
Though CEFORF-S is generally well-tolerated, some rare but significant side effects may occur. Prompt recognition and medical intervention are critical in such cases:
Hypersensitivity Reactions
- Rash, itching, fever, and in extreme cases, anaphylaxis.
- Always check for penicillin or cephalosporin allergy history.
Hematologic Effects
- Rare incidents of leukopenia, neutropenia, or thrombocytopenia.
- Monitor CBC in long-term or high-dose therapy.
Hepatic and Renal Issue
- Transient hepatic dysfunction, including elevated liver enzymes, is a possible side effect.
- Renal function monitoring is advisable in high-risk groups.
Superinfections
- Overuse may lead to Clostridium difficile-associated diarrhea or fungal overgrowth.
Local Injection Site Reactions
- Pain, redness, or hardening at the injection site, especially with IM route.
- IV route may cause phlebitis; rotate sites if prolonged use is required.
In all cases, discontinue use and consult a physician if any adverse effects arise. Supportive care and alternative antibiotics may be required.
Conclusion
CEFORF-S (375 mg, 750 mg, 1.5 g) is a potent combination of Ceftriaxone and Sulbactam, effectively targeting a broad spectrum of bacterial infections. It provides flexibility in dosage and administration, making it suitable across patient profiles—from mild outpatient cases to severe inpatient infections. While generally safe, clinicians should remain vigilant about rare but serious side effects. With responsible use under medical supervision, CEFORF-S remains a reliable choice in the battle against resistant infections. Always follow prescribed dosing guidelines and clinical protocols for optimal results.
